Functional Identification and Structural Analysis of a New Lipoate Protein Ligase inMycoplasma hyopneumoniae.
Zhu, K., Chen, H., Jin, J., Wang, N., Ma, G., Huang, J., Feng, Y., Xin, J., Zhang, H., Liu, H.(2020) Front Cell Infect Microbiol 10: 156-156
- PubMed: 32373550
- DOI: https://doi.org/10.3389/fcimb.2020.00156
- Primary Citation of Related Structures:
6JOM - PubMed Abstract:
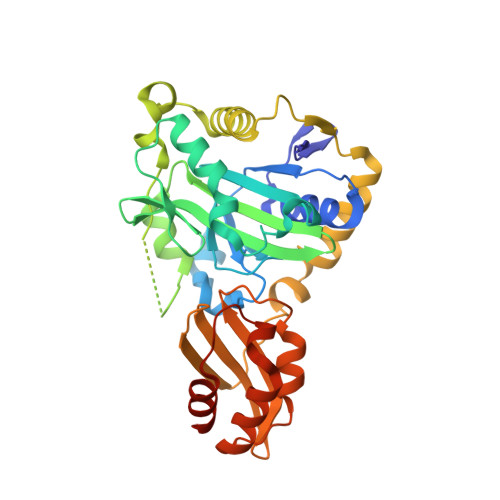
Mycoplasma hyopneumoniae ( M. hyopneumoniae ) is the causative agent of pandemic pneumonia among pigs, namely, swine enzootic pneumonia. Although M. hyopneumoniae was first identified in 1965, little is known regarding its metabolic pathways, which might play a pivotal role during disease pathogenesis. Lipoate is an essential cofactor for enzymes important for central metabolism. However, the lipoate metabolism pathway in M. hyopneumoniae is definitely unclear. Here, we identified a novel gene, lpl, encoding a lipoate protein ligase in the genome of M. hyopneumoniae (Mhp-Lpl). This gene contains 1,032 base pairs and encodes a protein of 343 amino acids, which is between 7.5 and 36.09% identical to lipoate protein ligases (Lpls) of other species. Similar to its homologs in other species, Mhp-Lpl catalyzes the ATP-dependent activation of lipoate to lipoyl-AMP and the transfer of the activated lipoyl onto the lipoyl domains of M. hyopneumoniae GcvH (Mhp H) in vitro . Enzymatic and mutagenesis analysis indicate that residue K56 within the SKT sequence of Mhp H protein is the lipoyl moiety acceptor site. The three-dimensional structure showed typical lipoate protein ligase folding, with a large N-terminal domain and a small C-terminal domain. The large N-terminal domain is responsible for the full enzymatic activity of Mhp-Lpl. The identification and characterization of Mhp-Lpl will be beneficial to our understanding of M. hyopneumoniae metabolism.
Organizational Affiliation:
State Key Laboratory of Veterinary Biotechnology, Harbin Veterinary Research Institute, The Chinese Academy of Agricultural Sciences, Harbin, China.