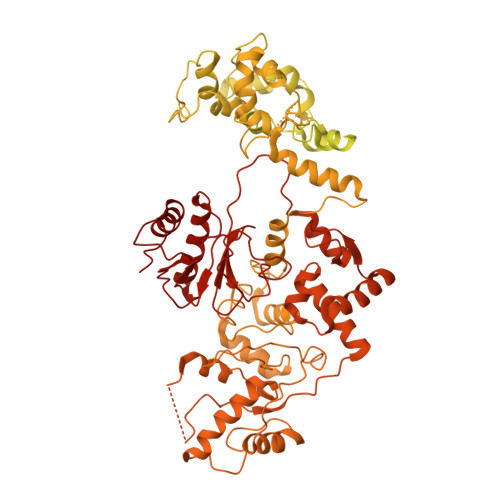
Oligomeric quaternary structure of Escherichia coli and Mycobacterium smegmatis Lhr helicases is nucleated by a novel C-terminal domain composed of five winged-helix modules.
Warren, G.M., Wang, J., Patel, D.J., Shuman, S.(2021) Nucleic Acids Res 49: 3876-3887
- PubMed: 33744958
- DOI: https://doi.org/10.1093/nar/gkab145
- Primary Citation of Related Structures:
7LHL - PubMed Abstract:
Mycobacterium smegmatis Lhr (MsmLhr; 1507-aa) is the founder of a novel clade of bacterial helicases. MsmLhr consists of an N-terminal helicase domain (aa 1-856) with a distinctive tertiary structure (Lhr-Core) and a C-terminal domain (Lhr-CTD) of unknown structure. Here, we report that Escherichia coli Lhr (EcoLhr; 1538-aa) is an ATPase, translocase and ATP-dependent helicase. Like MsmLhr, EcoLhr translocates 3' to 5' on ssDNA and unwinds secondary structures en route, with RNA:DNA hybrid being preferred versus DNA:DNA duplex. The ATPase and translocase activities of EcoLhr inhere to its 877-aa Core domain. Full-length EcoLhr and MsmLhr have homo-oligomeric quaternary structures in solution, whereas their respective Core domains are monomers. The MsmLhr CTD per se is a homo-oligomer in solution. We employed cryo-EM to solve the structure of the CTD of full-length MsmLhr. The CTD protomer is composed of a series of five winged-helix (WH) modules and a β-barrel module. The CTD adopts a unique homo-tetrameric quaternary structure. A Lhr-CTD subdomain, comprising three tandem WH modules and the β-barrel, is structurally homologous to AlkZ, a bacterial DNA glycosylase that recognizes and excises inter-strand DNA crosslinks. This homology is noteworthy given that Lhr is induced in mycobacteria exposed to the inter-strand crosslinker mitomycin C.
Organizational Affiliation:
Molecular Biology and Structural Biology Programs, Memorial Sloan Kettering Cancer Center, New York, NY 10065, USA.