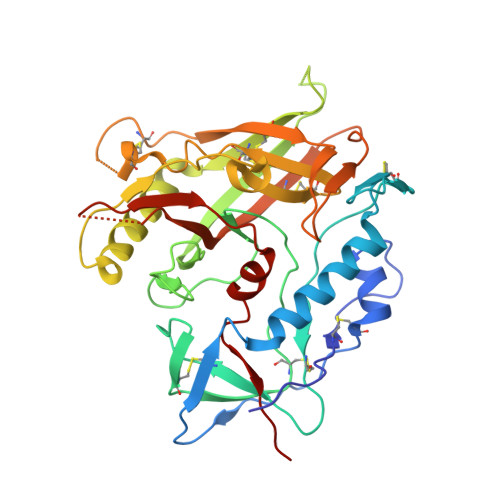
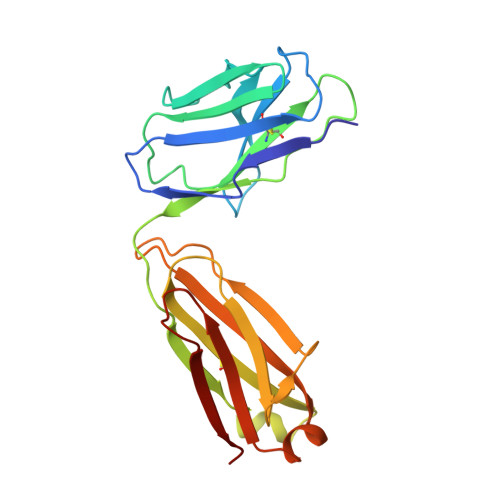
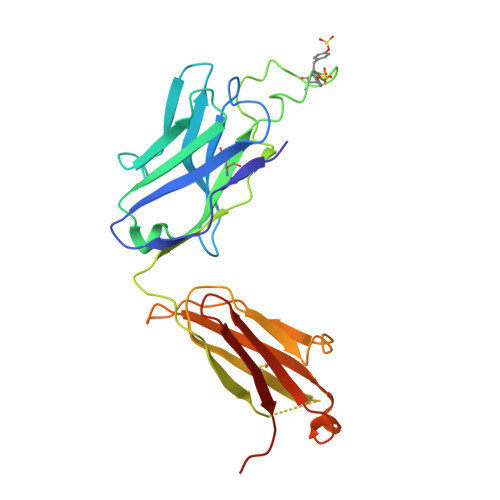
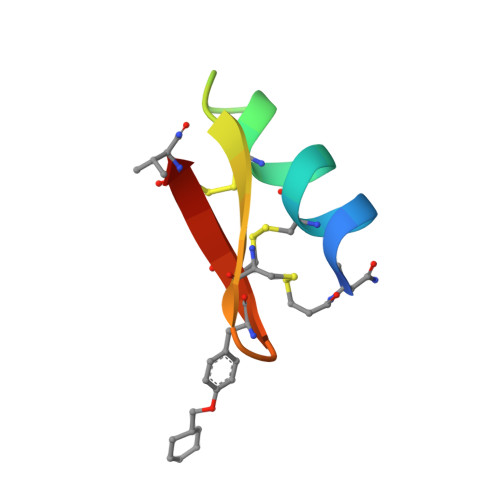
Defining rules governing recognition and Fc-mediated effector functions to the HIV-1 co-receptor binding site.
Tolbert, W.D., Sherburn, R., Gohain, N., Ding, S., Flinko, R., Orlandi, C., Ray, K., Finzi, A., Lewis, G.K., Pazgier, M.(2020) BMC Biol 18: 91-91
- PubMed: 32693837
- DOI: https://doi.org/10.1186/s12915-020-00819-y
- Primary Citation of Related Structures:
6W4M - PubMed Abstract:
The binding of HIV-1 Envelope glycoproteins (Env) to host receptor CD4 exposes vulnerable conserved epitopes within the co-receptor binding site (CoRBS) which are required for the engagement of either CCR5 or CXCR4 co-receptor to allow HIV-1 entry. Antibodies against this region have been implicated in the protection against HIV acquisition in non-human primate (NHP) challenge studies and found to act synergistically with antibodies of other specificities to deliver effective Fc-mediated effector function against HIV-1-infected cells. Here, we describe the structure and function of N12-i2, an antibody isolated from an HIV-1-infected individual, and show how the unique structural features of this antibody allow for its effective Env recognition and Fc-mediated effector function.
Organizational Affiliation:
Infectious Disease Division, Department of Medicine, Uniformed Services University of the Health Sciences, 4301 Jones Bridge Road, Bethesda, MD, 20814-4712, USA.