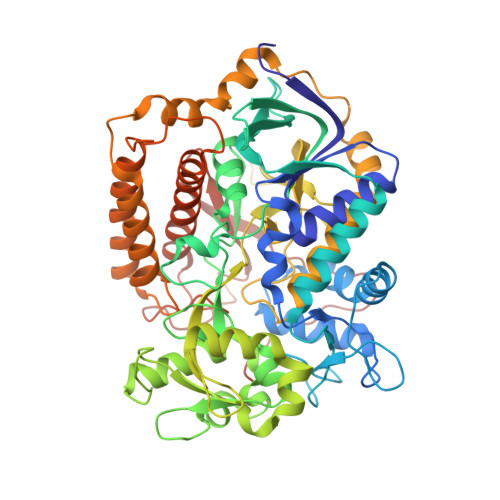
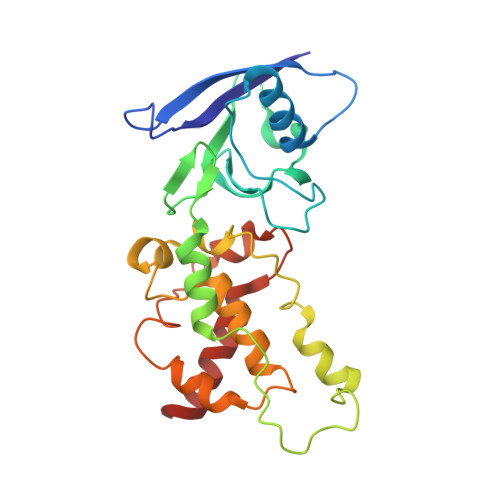
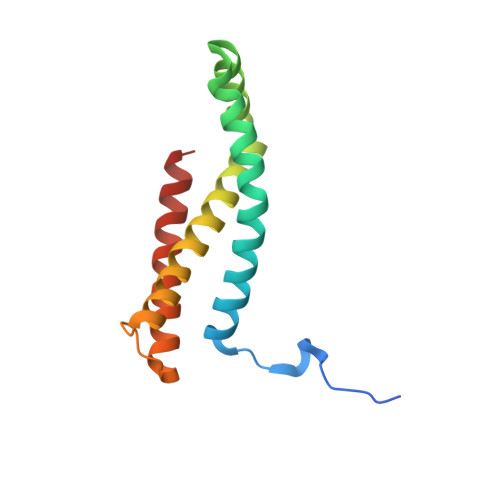
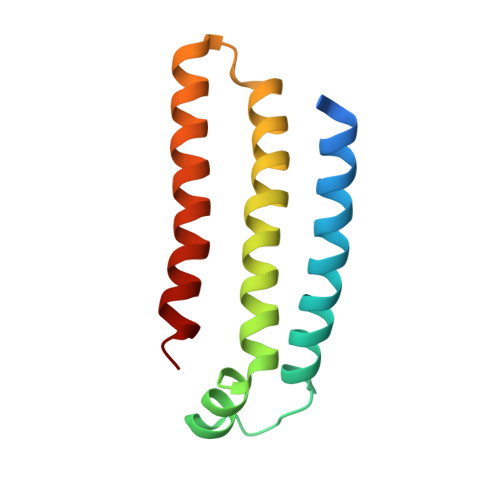
Structure of Escherichia Coli Succinate:Quinone Oxidoreductase with an Occupied and Empty Quinone- Binding Site.
Ruprecht, J., Yankovskaya, V., Maklashina, E., Iwata, S., Cecchini, G.(2009) J Biological Chem 284: 29836
- PubMed: 19710024 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1074/jbc.M109.010058
- Primary Citation Related Structures:
2WDQ, 2WDR, 2WDV - PubMed Abstract:
Three new structures of Escherichia coli succinate-quinone oxidoreductase (SQR) have been solved. One with the specific quinone-binding site (Q-site) inhibitor carboxin present has been solved at 2.4 A resolution and reveals how carboxin inhibits the Q-site. The other new structures are with the Q-site inhibitor pentachlorophenol and with an empty Q-site. These structures reveal important details unresolved in earlier structures. Comparison of the new SQR structures shows how subtle rearrangements of the quinone-binding site accommodate the different inhibitors. The position of conserved water molecules near the quinone binding pocket leads to a reassessment of possible water-mediated proton uptake networks that complete reduction of ubiquinone. The dicarboxylate-binding site in the soluble domain of SQR is highly similar to that seen in high resolution structures of avian SQR (PDB 2H88) and soluble flavocytochrome c (PDB 1QJD) showing mechanistically significant structural features conserved across prokaryotic and eukaryotic SQRs.
- Membrane Protein Crystallography Group, Molecular Biosciences Division, Imperial College, London SW72AZ, United Kingdom.
Organizational Affiliation: