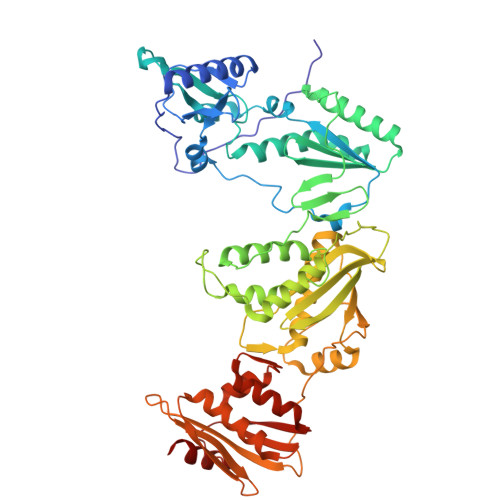
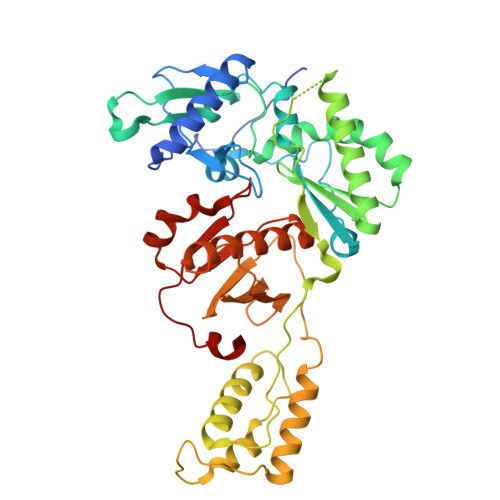
Snapshot of the equilibrium dynamics of a drug bound to HIV-1 reverse transcriptase.
Kuroda, D.G., Bauman, J.D., Challa, J.R., Patel, D., Troxler, T., Das, K., Arnold, E., Hochstrasser, R.M.(2013) Nat Chem 5: 174-181
- PubMed: 23422558 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/nchem.1559
- Primary Citation Related Structures:
4G1Q - PubMed Abstract:
The anti-AIDS drug rilpivirine undergoes conformational changes to bind HIV-1 reverse transcriptase (RT), which is an essential enzyme for the replication of HIV. These changes allow it to retain potency against mutations that otherwise would render the enzyme resistant. Here we report that water molecules play an essential role in this binding process. Femtosecond experiments and theory expose the molecular level dynamics of rilpivirine bound to HIV-1 RT. Two nitrile substituents, one on each arm of the drug, are used as vibrational probes of the structural dynamics within the binding pocket. Two-dimensional vibrational echo spectroscopy reveals that one nitrile group is unexpectedly hydrogen-bonded to a mobile water molecule, not identified in previous X-ray structures. Ultrafast nitrile-water dynamics are confirmed by simulations. A higher (1.51 Å) resolution X-ray structure also reveals a water-drug interaction network. Maintenance of a crucial anchoring hydrogen bond may help retain the potency of rilpivirine against pocket mutations despite the structural variations they cause.
- Ultrafast Optical Processes Laboratory, Department of Chemistry, University of Pennsylvania, Pennsylvania 19067, USA.
Organizational Affiliation: