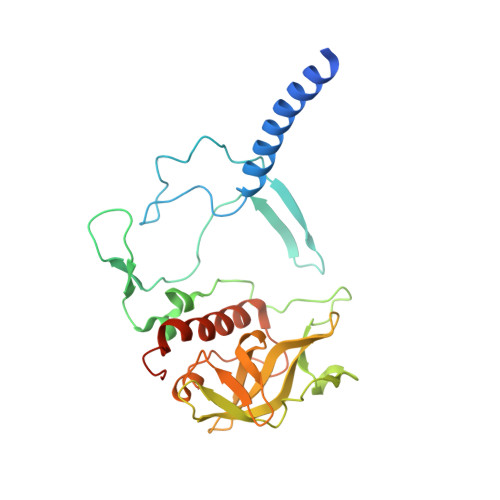
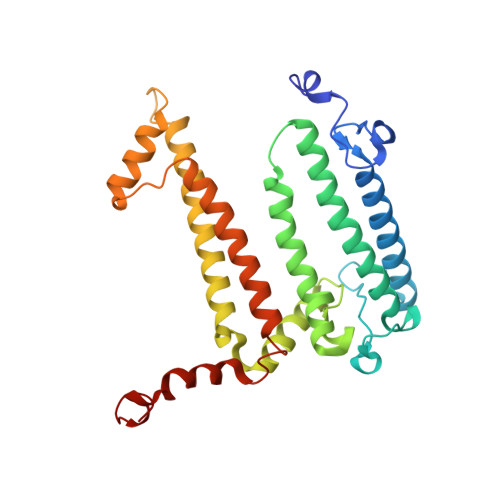
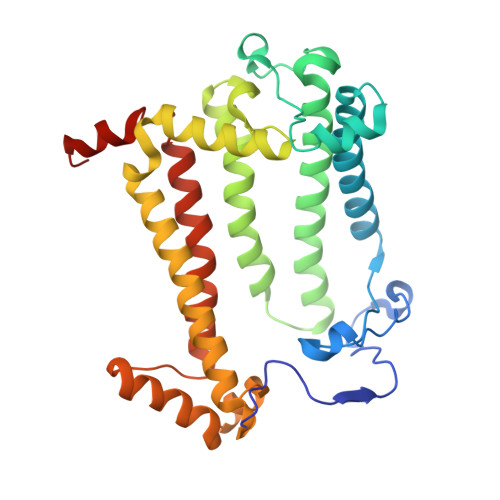
Role of Rhodobacter sphaeroides photosynthetic reaction center residue M214 in the composition, absorbance properties, and conformations of H(A) and B(A) cofactors.
Saer, R.G., Hardjasa, A., Rosell, F.I., Mauk, A.G., Murphy, M.E., Beatty, J.T.(2013) Biochemistry 52: 2206-2217
- PubMed: 23480277 Search on PubMed
- DOI: https://doi.org/10.1021/bi400207m
- Primary Citation Related Structures:
4IN5, 4IN6, 4IN7 - PubMed Abstract:
In the native reaction center (RC) of Rhodobacter sphaeroides, the side chain of (M)L214 projects orthogonally toward the plane and into the center of the A branch bacteriopheophytin (BPhe) macrocycle. The possibility that this side chain is responsible for the dechelation of the central Mg(2+) of bacteriochlorophyll (BChl) was investigated by replacement of (M)214 with residues possessing small, nonpolar side chains that can neither coordinate nor block access to the central metal ion. The (M)L214 side chain was also replaced with Cys, Gln, and Asn to evaluate further the requirements for assembly of the RC with BChl in the HA pocket. Photoheterotrophic growth studies showed no difference in growth rates of the (M)214 nonpolar mutants at a low light intensity, but the growth of the amide-containing mutants was impaired. The absorbance spectra of purified RCs indicated that although absorbance changes are associated with the nonpolar mutations, the nonpolar mutant RC pigment compositions are the same as in the wild-type protein. Crystal structures of the (M)L214G, (M)L214A, and (M)L214N mutants were determined (determined to 2.2-2.85 Å resolution), confirming the presence of BPhe in the HA pocket and revealing alternative conformations of the phytyl tail of the accessory BChl in the BA site of these nonpolar mutants. Our results demonstrate that (i) BChl is converted to BPhe in a manner independent of the aliphatic side chain length of nonpolar residues replacing (M)214, (ii) BChl replaces BPhe if residue (M)214 has an amide-bearing side chain, (iii) (M)214 side chains containing sulfur are not sufficient to bind BChl in the HA pocket, and (iv) the (M)214 side chain influences the conformation of the phytyl tail of the BA BChl.
- Department of Microbiology and Immunology, The University of British Columbia, 2350 Health Sciences Mall, Vancouver, BC, Canada V6T 1Z3.
Organizational Affiliation: