A structure of the GEF Kalirin DH1 domain in complex with the small GTPase Rac1
Gray, J., Krojer, T., Talon, R., Fairhead, M., Bountra, C., Arrowsmith, C.H., Edwards, A., Brennan, P., von Delft, F.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Ras-related C3 botulinum toxin substrate 1 | 177 | Homo sapiens | Mutation(s): 0 Gene Names: RAC1, TC25, MIG5 EC: 3.6.5.2 | 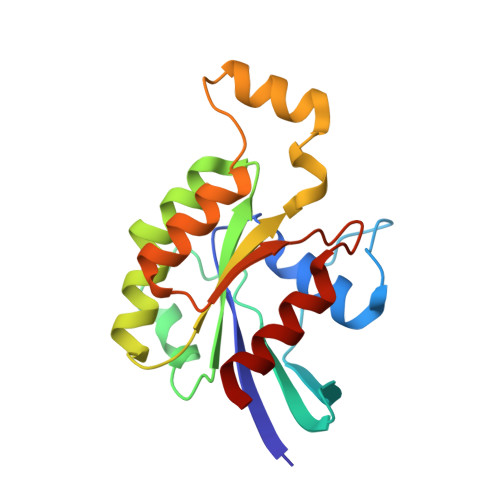 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P63000 GTEx: ENSG00000136238 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P63000 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Kalirin | 180 | Rattus norvegicus | Mutation(s): 0 Gene Names: Kalrn, Duo, Hapip EC: 2.7.11.1 | 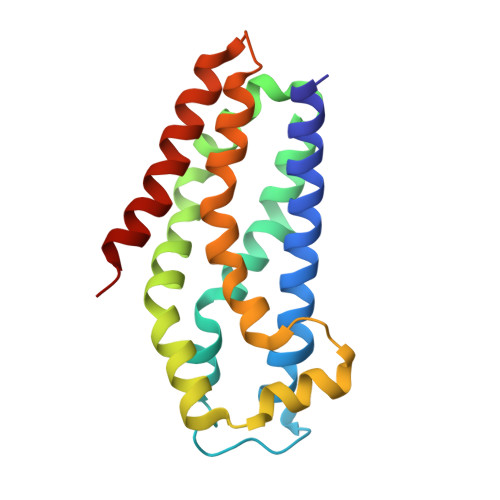 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P97924 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| GDP Download:Ideal Coordinates CCD File | E [auth A] | GUANOSINE-5'-DIPHOSPHATE C10 H15 N5 O11 P2 QGWNDRXFNXRZMB-UUOKFMHZSA-N |  | ||
| EDO Download:Ideal Coordinates CCD File | C [auth A] D [auth A] F [auth B] G [auth B] H [auth B] | 1,2-ETHANEDIOL C2 H6 O2 LYCAIKOWRPUZTN-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 63.255 | α = 90 |
| b = 63.255 | β = 90 |
| c = 346.704 | γ = 120 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| xia2 | data reduction |
| xia2 | data scaling |
| PHENIX | phasing |