GLMP is essential for bone-marrow hematopoiesis and lysosomal glycolipid metabolism
Manzanillo, P., Huang, C.S., Boenig, G., Calses, P., Scherl, A., Reichelt, M., Katakam, A.K., Martin, F., Hymowitz, S.G., Ouyang, W.To be published.
Experimental Data Snapshot
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| 1H3 Fab light chain | A [auth L] | 214 | Mus musculus | Mutation(s): 0 | 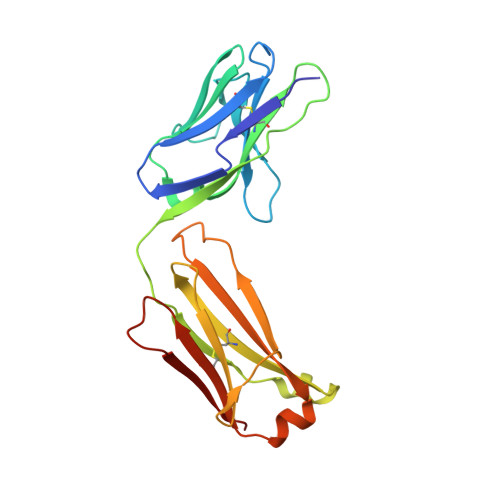 |
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| 1H3 Fab heavy chain | B [auth H] | 227 | Mus musculus | Mutation(s): 0 | 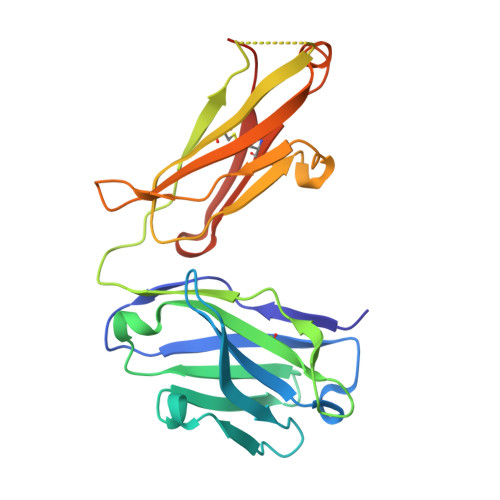 |
Entity ID: 3 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Glycosylated lysosomal membrane protein | 404 | Mus musculus | Mutation(s): 0 Gene Names: Glmp | 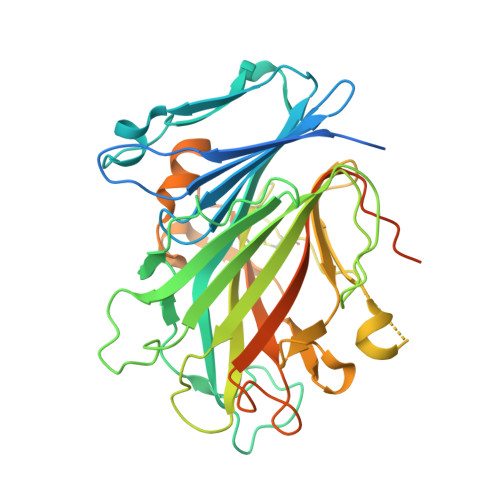 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q9JHJ3 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| NAG Download:Ideal Coordinates CCD File | H [auth C] I [auth C] J [auth C] K [auth C] L [auth C] | 2-acetamido-2-deoxy-beta-D-glucopyranose C8 H15 N O6 OVRNDRQMDRJTHS-FMDGEEDCSA-N |  | ||
| NA Download:Ideal Coordinates CCD File | D [auth H], E [auth C], F [auth C], G [auth C] | SODIUM ION Na FKNQFGJONOIPTF-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 81.924 | α = 90 |
| b = 55.238 | β = 99.35 |
| c = 99.639 | γ = 90 |
| Software Name | Purpose |
|---|---|
| REFMAC | refinement |
| HKL-2000 | data reduction |
| HKL-2000 | data scaling |
| PHASER | phasing |