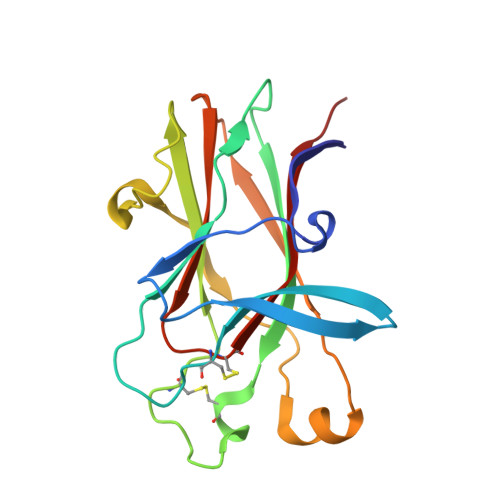
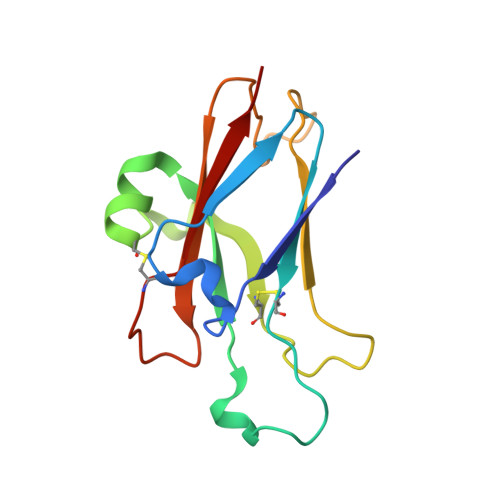
Ligand recognition by A-class Eph receptors: crystal structures of the EphA2 ligand-binding domain and the EphA2/ephrin-A1 complex.
Himanen, J.P., Goldgur, Y., Miao, H., Myshkin, E., Guo, H., Buck, M., Nguyen, M., Rajashankar, K.R., Wang, B., Nikolov, D.B.(2009) EMBO Rep 10: 722-728
- PubMed: 19525919 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/embor.2009.91
- Primary Citation Related Structures:
3HEI, 3HPN - PubMed Abstract:
Ephrin (Eph) receptor tyrosine kinases fall into two subclasses (A and B) according to preferences for their ephrin ligands. All published structural studies of Eph receptor/ephrin complexes involve B-class receptors. Here, we present the crystal structures of an A-class complex between EphA2 and ephrin-A1 and of unbound EphA2. Although these structures are similar overall to their B-class counterparts, they reveal important differences that define subclass specificity. The structures suggest that the A-class Eph receptor/ephrin interactions involve smaller rearrangements in the interacting partners, better described by a 'lock-and-key'-type binding mechanism, in contrast to the 'induced fit' mechanism defining the B-class molecules. This model is supported by structure-based mutagenesis and by differential requirements for ligand oligomerization by the two subclasses in cell-based Eph receptor activation assays. Finally, the structure of the unligated receptor reveals a homodimer assembly that might represent EphA2-specific homotypic cell adhesion interactions.
- Structural Biology Program, Memorial Sloan-Kettering Cancer Center, New York, New York 10021, USA.
Organizational Affiliation: