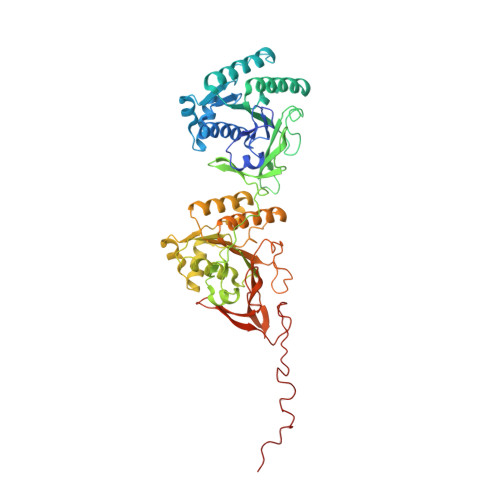
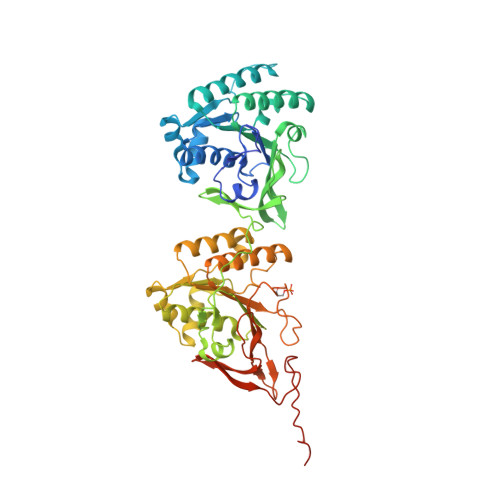
Structures of KaiC Circadian Clock Mutant Proteins: A New Phosphorylation Site at T426 and Mechanisms of Kinase, ATPase and Phosphatase.
Pattanayek, R., Mori, T., Xu, Y., Pattanayek, S., Johnson, C.H., Egli, M.(2009) PLoS One 4: e7529-e7529
- PubMed: 19956664 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1371/journal.pone.0007529
- Primary Citation Related Structures:
3JZM, 3K09, 3K0A, 3K0C, 3K0E, 3K0F - PubMed Abstract:
The circadian clock of the cyanobacterium Synechococcus elongatus can be reconstituted in vitro by three proteins, KaiA, KaiB and KaiC. Homo-hexameric KaiC displays kinase, phosphatase and ATPase activities; KaiA enhances KaiC phosphorylation and KaiB antagonizes KaiA. Phosphorylation and dephosphorylation of the two known sites in the C-terminal half of KaiC subunits, T432 and S431, follow a strict order (TS-->pTS-->pTpS-->TpS-->TS) over the daily cycle, the origin of which is not understood. To address this void and to analyze the roles of KaiC active site residues, in particular T426, we determined structures of single and double P-site mutants of S. elongatus KaiC. The conformations of the loop region harboring P-site residues T432 and S431 in the crystal structures of six KaiC mutant proteins exhibit subtle differences that result in various distances between Thr (or Ala/Asn/Glu) and Ser (or Ala/Asp) residues and the ATP gamma-phosphate. T432 is phosphorylated first because it lies consistently closer to Pgamma. The structures of the S431A and T432E/S431A mutants reveal phosphorylation at T426. The environments of the latter residue in the structures and functional data for T426 mutants in vitro and in vivo imply a role in dephosphorylation. We provide evidence for a third phosphorylation site in KaiC at T426. T426 and S431 are closely spaced and a KaiC subunit cannot carry phosphates at both sites simultaneously. Fewer subunits are phosphorylated at T426 in the two KaiC mutants compared to phosphorylated T432 and/or S431 residues in the structures of wt and other mutant KaiCs, suggesting that T426 phosphorylation may be labile. The structures combined with functional data for a host of KaiC mutant proteins help rationalize why S431 trails T432 in the loss of its phosphate and shed light on the mechanisms of the KaiC kinase, ATPase and phosphatase activities.
- Department of Biochemistry, Vanderbilt University, School of Medicine, Nashville, Tennessee, United States of America.
Organizational Affiliation: