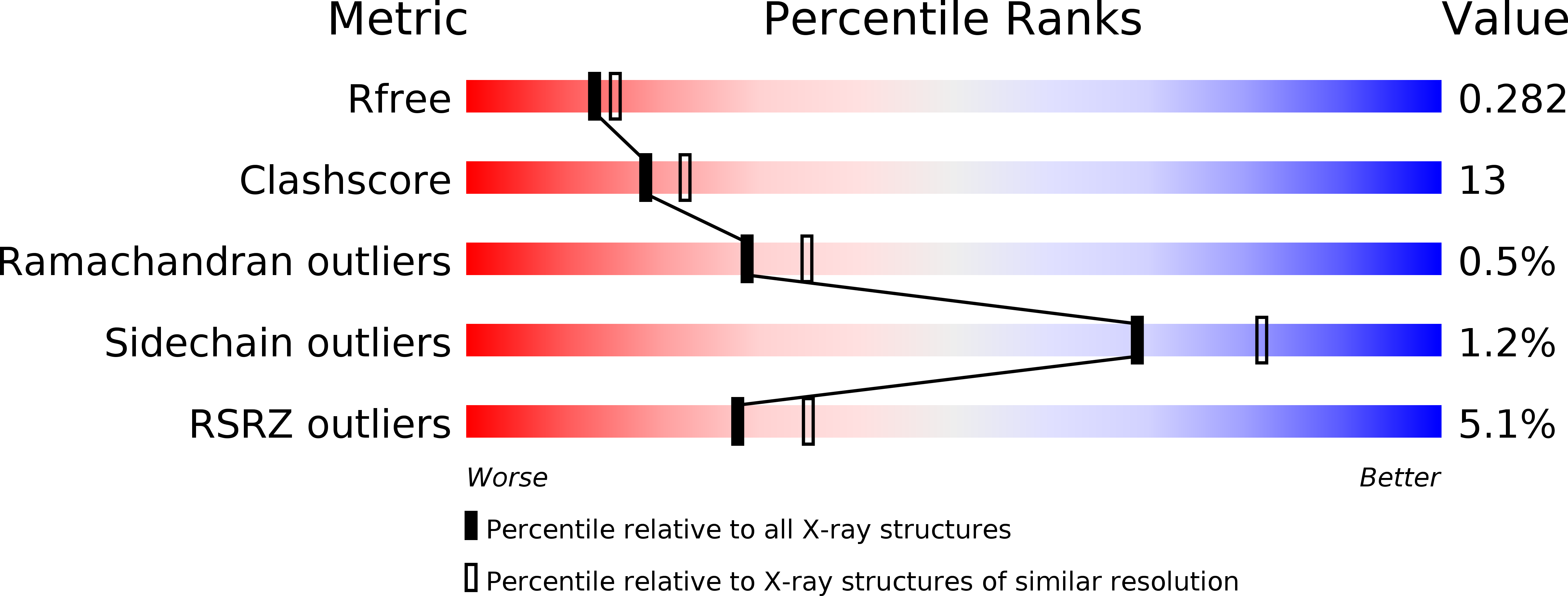
Biochemical, structural and molecular dynamics analyses of the potential virulence factor RipA from Yersinia pestis.
Torres, R., Swift, R.V., Chim, N., Wheatley, N., Lan, B., Atwood, B.R., Pujol, C., Sankaran, B., Bliska, J.B., Amaro, R.E., Goulding, C.W.(2011) PLoS One 6: e25084-e25084
- PubMed: 21966419
- DOI: https://doi.org/10.1371/journal.pone.0025084
- Primary Citation of Related Structures:
3QLI, 3QLK, 3S8D - PubMed Abstract:
Human diseases are attributed in part to the ability of pathogens to evade the eukaryotic immune systems. A subset of these pathogens has developed mechanisms to survive in human macrophages. Yersinia pestis, the causative agent of the bubonic plague, is a predominately extracellular pathogen with the ability to survive and replicate intracellularly. A previous study has shown that a novel rip (required for intracellular proliferation) operon (ripA, ripB and ripC) is essential for replication and survival of Y. pestis in postactivated macrophages, by playing a role in lowering macrophage-produced nitric oxide (NO) levels. A bioinformatics analysis indicates that the rip operon is conserved among a distally related subset of macrophage-residing pathogens, including Burkholderia and Salmonella species, and suggests that this previously uncharacterized pathway is also required for intracellular survival of these pathogens. The focus of this study is ripA, which encodes for a protein highly homologous to 4-hydroxybutyrate-CoA transferase; however, biochemical analysis suggests that RipA functions as a butyryl-CoA transferase. The 1.9 Å X-ray crystal structure reveals that RipA belongs to the class of Family I CoA transferases and exhibits a unique tetrameric state. Molecular dynamics simulations are consistent with RipA tetramer formation and suggest a possible gating mechanism for CoA binding mediated by Val227. Together, our structural characterization and molecular dynamic simulations offer insights into acyl-CoA specificity within the active site binding pocket, and support biochemical results that RipA is a butyryl-CoA transferase. We hypothesize that the end product of the rip operon is butyrate, a known anti-inflammatory, which has been shown to lower NO levels in macrophages. Thus, the results of this molecular study of Y. pestis RipA provide a structural platform for rational inhibitor design, which may lead to a greater understanding of the role of RipA in this unique virulence pathway.
Organizational Affiliation:
Department of Molecular Biology and Biochemistry, University of California Irvine, Irvine, California, United States of America.