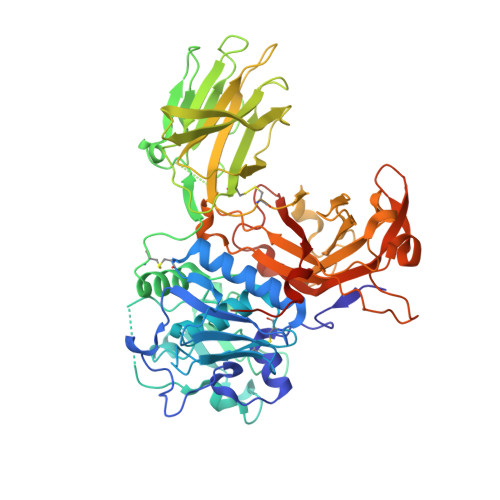
Structural basis for the sheddase function of human meprin beta metalloproteinase at the plasma membrane
Arolas, J.L., Broder, C., Jefferson, T., Guevara, T., Sterchi, E.E., Bode, W., Stocker, W., Becker-Pauly, C., Gomis-Ruth, F.X.(2012) Proc Natl Acad Sci U S A 109: 16131-16136
- PubMed: 22988105 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.1211076109
- Primary Citation Related Structures:
4GWM, 4GWN - PubMed Abstract:
Ectodomain shedding at the cell surface is a major mechanism to regulate the extracellular and circulatory concentration or the activities of signaling proteins at the plasma membrane. Human meprin β is a 145-kDa disulfide-linked homodimeric multidomain type-I membrane metallopeptidase that sheds membrane-bound cytokines and growth factors, thereby contributing to inflammatory diseases, angiogenesis, and tumor progression. In addition, it cleaves amyloid precursor protein (APP) at the β-secretase site, giving rise to amyloidogenic peptides. We have solved the X-ray crystal structure of a major fragment of the meprin β ectoprotein, the first of a multidomain oligomeric transmembrane sheddase, and of its zymogen. The meprin β dimer displays a compact shape, whose catalytic domain undergoes major rearrangement upon activation, and reveals an exosite and a sugar-rich channel, both of which possibly engage in substrate binding. A plausible structure-derived working mechanism suggests that substrates such as APP are shed close to the plasma membrane surface following an "N-like" chain trace.
- Proteolysis Laboratory, Department of Structural Biology, Molecular Biology Institute of Barcelona, Consejo Superior de Investigaciones Cientificas, Barcelona Science Park, E-08028 Barcelona, Spain.
Organizational Affiliation: