Structure, inhibition and regulation of two-pore channel TPC1 from Arabidopsis thaliana
Kintzer, A.F., Stroud, R.M.(2016) Nature 531: 258-262
Experimental Data Snapshot
(2016) Nature 531: 258-262
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Two pore calcium channel protein 1 | 723 | Arabidopsis thaliana | Mutation(s): 0 Gene Names: TPC1, CCH1, FOU2, At4g03560, F9H3.19, T5L23.5 Membrane Entity: Yes | 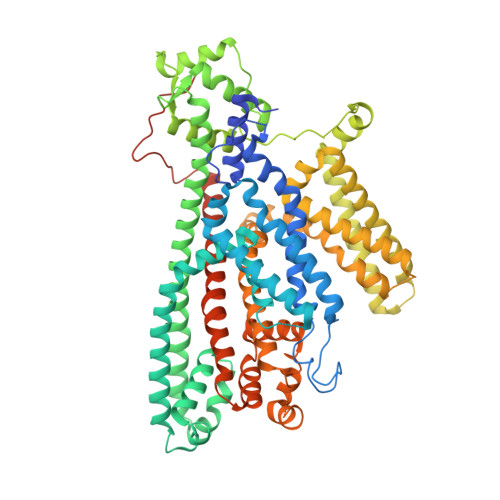 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q94KI8 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 3 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| 66R Download:Ideal Coordinates CCD File | J [auth A] | trans-Ned 19 C30 H31 F N4 O3 FUHCEERDBRGPQZ-LBNVMWSVSA-N |  | ||
| PLM Download:Ideal Coordinates CCD File | I [auth A] | PALMITIC ACID C16 H32 O2 IPCSVZSSVZVIGE-UHFFFAOYSA-N |  | ||
| CA Download:Ideal Coordinates CCD File | B [auth A] C [auth A] D [auth A] E [auth A] F [auth A] | CALCIUM ION Ca BHPQYMZQTOCNFJ-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 88.18 | α = 90 |
| b = 154.81 | β = 90 |
| c = 219.77 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| SHARP | phasing |
| DM | phasing |
| XDS | data reduction |
| XSCALE | data scaling |
| Funding Organization | Location | Grant Number |
|---|---|---|
| National Institutes of Health/National Institute of General Medical Sciences (NIH/NIGMS) | United States | GM24485 |