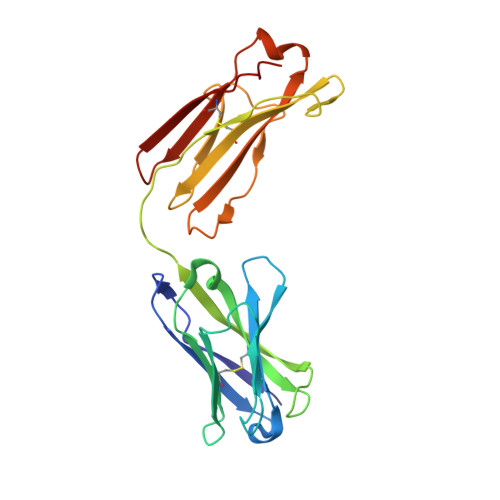
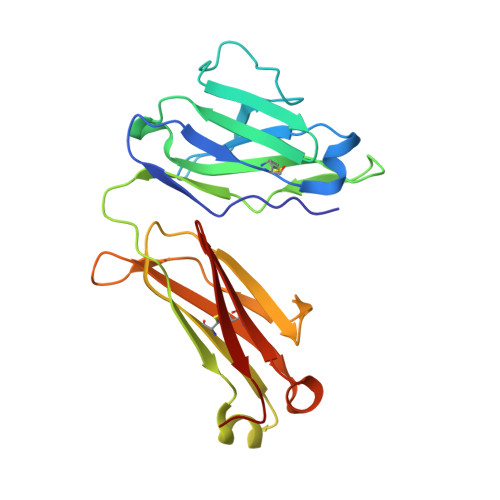
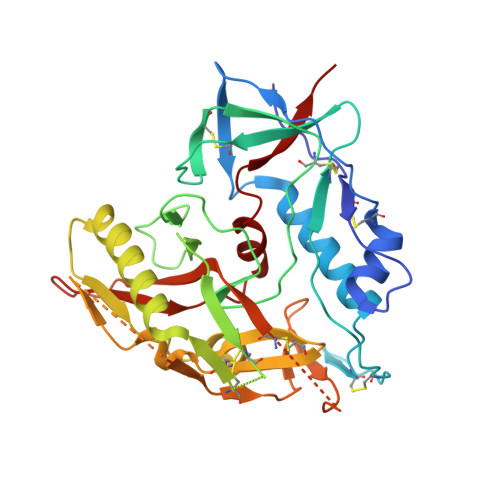
Structure of an N276-Dependent HIV-1 Neutralizing Antibody Targeting a Rare V5 Glycan Hole Adjacent to the CD4 Binding Site.
Wibmer, C.K., Gorman, J., Anthony, C.S., Mkhize, N.N., Druz, A., York, T., Schmidt, S.D., Labuschagne, P., Louder, M.K., Bailer, R.T., Abdool Karim, S.S., Mascola, J.R., Williamson, C., Moore, P.L., Kwong, P.D., Morris, L.(2016) J Virol 90: 10220-10235
- PubMed: 27581986 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1128/JVI.01357-16
- Primary Citation Related Structures:
5T33 - PubMed Abstract:
All HIV-1-infected individuals develop strain-specific neutralizing antibodies to their infecting virus, which in some cases mature into broadly neutralizing antibodies. Defining the epitopes of strain-specific antibodies that overlap conserved sites of vulnerability might provide mechanistic insights into how broadly neutralizing antibodies arise. We previously described an HIV-1 clade C-infected donor, CAP257, who developed broadly neutralizing plasma antibodies targeting an N276 glycan-dependent epitope in the CD4 binding site. The initial CD4 binding site response potently neutralized the heterologous tier 2 clade B viral strain RHPA, which was used to design resurfaced gp120 antigens for single-B-cell sorting. Here we report the isolation and structural characterization of CAP257-RH1, an N276 glycan-dependent CD4 binding site antibody representative of the early CD4 binding site plasma response in donor CAP257. The cocrystal structure of CAP257-RH1 bound to RHPA gp120 revealed critical interactions with the N276 glycan, loop D, and V5, but not with aspartic acid 368, similarly to HJ16 and 179NC75. The CAP257-RH1 monoclonal antibody was derived from the immunoglobulin-variable IGHV3-33 and IGLV3-10 genes and neutralized RHPA but not the transmitted/founder virus from donor CAP257. Its narrow neutralization breadth was attributed to a binding angle that was incompatible with glycosylated V5 loops present in almost all HIV-1 strains, including the CAP257 transmitted/founder virus. Deep sequencing of autologous CAP257 viruses, however, revealed minority variants early in infection that lacked V5 glycans. These glycan-free V5 loops are unusual holes in the glycan shield that may have been necessary for initiating this N276 glycan-dependent CD4 binding site B-cell lineage. The conserved CD4 binding site on gp120 is a major target for HIV-1 vaccine design, but key events in the elicitation and maturation of different antibody lineages to this site remain elusive. Studies have shown that strain-specific antibodies can evolve into broadly neutralizing antibodies or in some cases act as helper lineages. Therefore, characterizing the epitopes of strain-specific antibodies may help to inform the design of HIV-1 immunogens to elicit broadly neutralizing antibodies. In this study, we isolate a narrowly neutralizing N276 glycan-dependent antibody and use X-ray crystallography and viral deep sequencing to describe how gp120 lacking glycans in V5 might have elicited these early glycan-dependent CD4 binding site antibodies. These data highlight how glycan holes can play a role in the elicitation of B-cell lineages targeting the CD4 binding site.
- Centre for HIV and STIs, National Institute for Communicable Diseases, National Health Laboratory Service, Johannesburg, South Africa.
Organizational Affiliation: