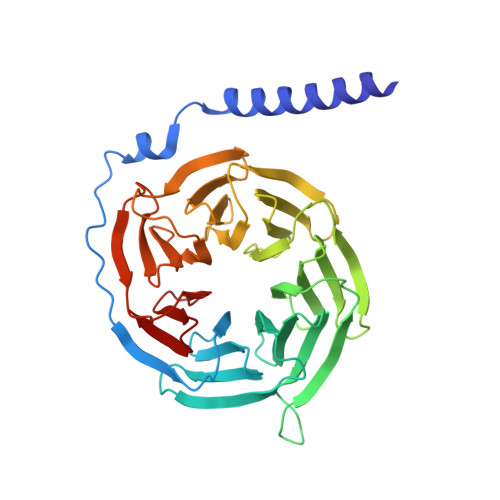
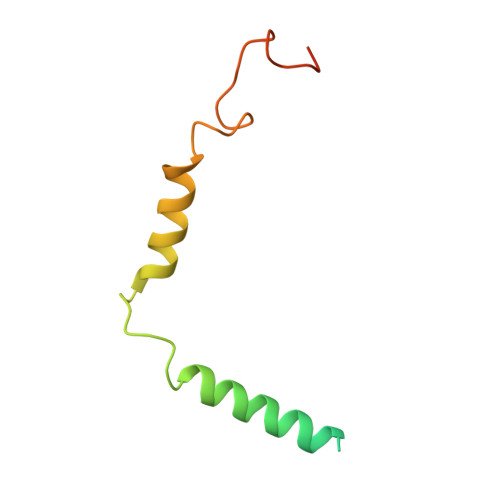
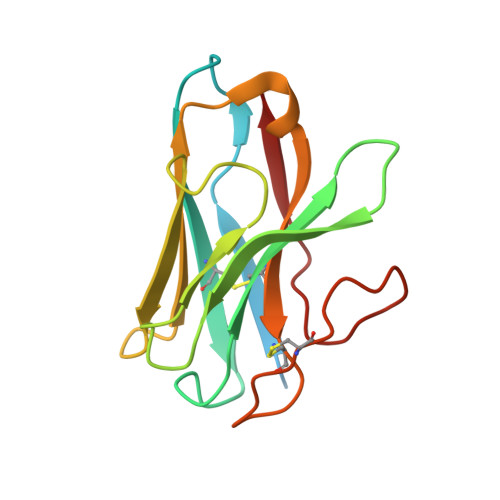
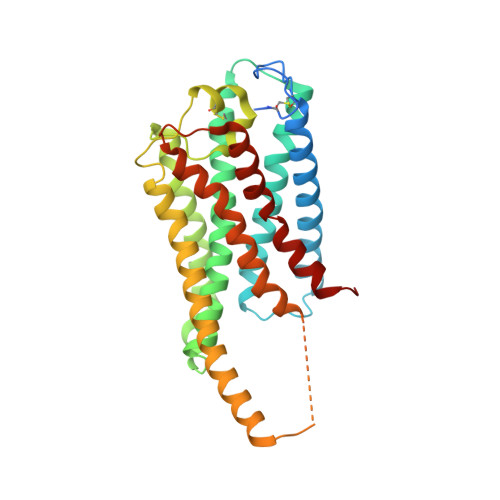
Allosteric coupling between G-protein binding and extracellular ligand binding sites in GPR52 revealed by 19 F-NMR and cryo-electron microscopy.
Fan, Y., Lin, X., Pan, B., Chen, B., Liu, D., Wuthrich, K., Xu, F.(2023) MedComm (2020) 4: e260-e260
- PubMed: 37051308 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1002/mco2.260
- Primary Citation Related Structures:
8HMP - iHuman Institute School of Life Science and Technology Shanghai Key Laboratory of High-resolution Electron Microscopy ShanghaiTech University Shanghai China.
Organizational Affiliation: