Cryo-EM structure of the full-length alpha1beta3gamma2 GABA(A) receptor in Saposin A nanodisc bound to GABA and Etomidate in the long-lived symmetric desensitised state
Mihaylov, D.B., Malinauskas, T., Aricescu, A.R.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Gamma-aminobutyric acid receptor subunit alpha-1 | 464 | Homo sapiens | Mutation(s): 0 Gene Names: GABRA1 | 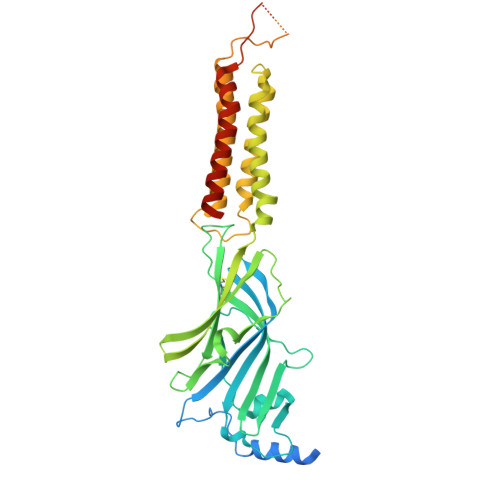 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P14867 GTEx: ENSG00000022355 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P14867 | ||||
Glycosylation | |||||
| Glycosylation Sites: 1 | Go to GlyGen: P14867-1 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Gamma-aminobutyric acid receptor subunit beta-3 | 473 | Homo sapiens | Mutation(s): 0 Gene Names: GABRB3 | 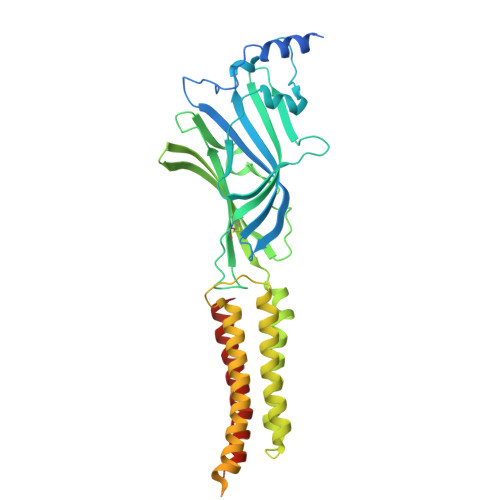 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P28472 GTEx: ENSG00000166206 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P28472 | ||||
Glycosylation | |||||
| Glycosylation Sites: 2 | Go to GlyGen: P28472-1 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 3 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Gamma-aminobutyric acid receptor subunit gamma-2 | 495 | Homo sapiens | Mutation(s): 0 Gene Names: GABRG2 | 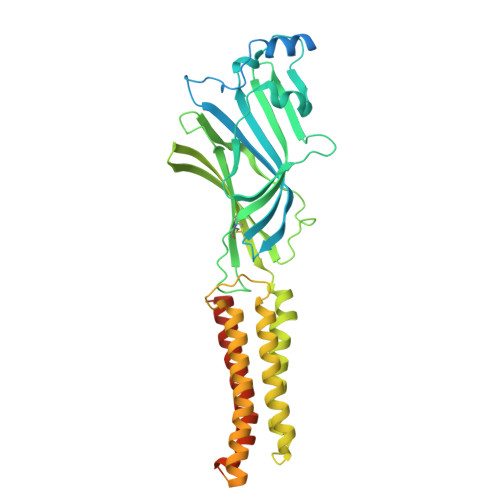 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P18507 GTEx: ENSG00000113327 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P18507 | ||||
Glycosylation | |||||
| Glycosylation Sites: 1 | Go to GlyGen: P18507-2 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 4 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Megabody-38 | F [auth G] | 133 | Lama glama | Mutation(s): 0 | 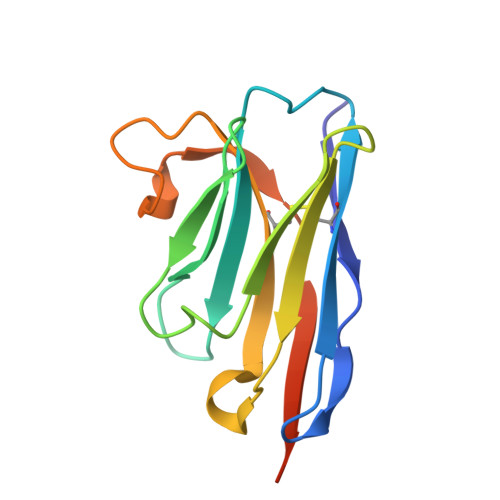 |
Entity ID: 5 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Length | 2D Diagram | Glycosylation | D Interactions |
| alpha-D-mannopyranose-(1-2)-alpha-D-mannopyranose-(1-2)-alpha-D-mannopyranose-(1-3)-[alpha-D-mannopyranose-(1-3)-[alpha-D-mannopyranose-(1-6)]alpha-D-mannopyranose-(1-6)]beta-D-mannopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose | G [auth F] | 9 |  | N-Glycosylation | |
Glycosylation Resources | |||||
GlyTouCan: G68668TB GlyCosmos: G68668TB GlyGen: G68668TB | |||||
Entity ID: 6 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Length | 2D Diagram | Glycosylation | D Interactions |
| 2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose | H, K | 2 |  | N-Glycosylation | |
Glycosylation Resources | |||||
GlyTouCan: G42666HT GlyCosmos: G42666HT GlyGen: G42666HT | |||||
Entity ID: 7 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Length | 2D Diagram | Glycosylation | D Interactions |
| alpha-D-mannopyranose-(1-3)-alpha-D-mannopyranose-(1-6)-[alpha-D-mannopyranose-(1-3)]beta-D-mannopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose | I, L | 6 |  | N-Glycosylation | |
Glycosylation Resources | |||||
GlyTouCan: G09724ZC GlyCosmos: G09724ZC GlyGen: G09724ZC | |||||
Entity ID: 8 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Length | 2D Diagram | Glycosylation | D Interactions |
| alpha-D-mannopyranose-(1-3)-[alpha-D-mannopyranose-(1-6)]beta-D-mannopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose | J | 5 |  | N-Glycosylation | |
Glycosylation Resources | |||||
GlyTouCan: G22768VO GlyCosmos: G22768VO GlyGen: G22768VO | |||||
| Ligands 7 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| PIO Download:Ideal Coordinates CCD File | M [auth A], V [auth D] | [(2R)-2-octanoyloxy-3-[oxidanyl-[(1R,2R,3S,4R,5R,6S)-2,3,6-tris(oxidanyl)-4,5-diphosphonooxy-cyclohexyl]oxy-phosphoryl]oxy-propyl] octanoate C25 H49 O19 P3 XLNCEHRXXWQMPK-MJUMVPIBSA-N |  | ||
| V8D (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | BA [auth E], Q [auth B] | Etomidate C14 H16 N2 O2 NPUKDXXFDDZOKR-LLVKDONJSA-N |  | ||
| R16 Download:Ideal Coordinates CCD File | AA [auth E] | HEXADECANE C16 H34 DCAYPVUWAIABOU-UHFFFAOYSA-N |  | ||
| NAG Download:Ideal Coordinates CCD File | S [auth C] | 2-acetamido-2-deoxy-beta-D-glucopyranose C8 H15 N O6 OVRNDRQMDRJTHS-FMDGEEDCSA-N |  | ||
| D10 Download:Ideal Coordinates CCD File | N [auth A], R [auth B], T [auth C], W [auth D], Z [auth E] | DECANE C10 H22 DIOQZVSQGTUSAI-UHFFFAOYSA-N |  | ||
| ABU (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | P [auth B], Y [auth E] | GAMMA-AMINO-BUTANOIC ACID C4 H9 N O2 BTCSSZJGUNDROE-UHFFFAOYSA-N |  | ||
| CL Download:Ideal Coordinates CCD File | O [auth A], U [auth C], X [auth D] | CHLORIDE ION Cl VEXZGXHMUGYJMC-UHFFFAOYSA-M |  | ||
| Task | Software Package | Version |
|---|---|---|
| MODEL REFINEMENT | PHENIX | 1.21_5207 |
| RECONSTRUCTION | RELION |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Medical Research Council (MRC, United Kingdom) | United Kingdom | MC_UP_1201/15 |
| Cancer Research UK | United Kingdom | DRCRPG-May23/100002 |
| Medical Research Council (MRC, United Kingdom) | United Kingdom | MR/L009609/1 |
| Medical Research Council (MRC, United Kingdom) | United Kingdom | MC_EX_MR/T046279/1 |
| National Institutes of Health/National Institute of General Medical Sciences (NIH/NIGMS) | United States | 1R01-GM135550 |
| National Science Foundation (NSF, United States) | United States | NeuroNex 2014862 |