Integrative modelling reveals the structure of the human Mic60-Mic19 subcomplex and its role as a diffusion barrier in mitochondria
Nathanail, E., Rolando, E., Ruwolt, M., Zaporozhets, I., Liu, F., Clementi, C., Daumke, O.(2026) bioRxiv
Experimental Data Snapshot
Starting Model: in silico
View more details
wwPDB Validation 3D Report Full Report
(2026) bioRxiv
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| MICOS complex subunit MIC60 | 183 | Homo sapiens | Mutation(s): 0 Gene Names: IMMT, HMP, MIC60, MINOS2, PIG4, PIG52 | 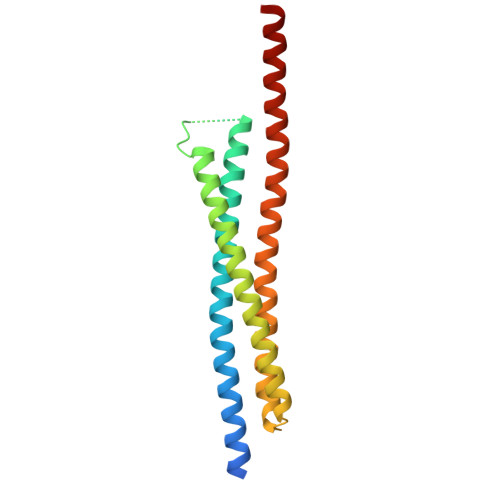 | |
UniProt & NIH Common Fund Data Resources | |||||
Find proteins for Q16891 (Homo sapiens) Explore Q16891 Go to UniProtKB: Q16891 | |||||
PHAROS: Q16891 GTEx: ENSG00000132305 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q16891 | ||||
Sequence AnnotationsExpand | |||||
| |||||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 76.12 | α = 90 |
| b = 76.12 | β = 90 |
| c = 168.41 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| XSCALE | data scaling |
| XDS | data reduction |
| PHASER | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Helmholtz Association | Germany | -- |