Crystal complex of murine cycloxygenase-2 with alpaca nanobody F9
Xu, S., Uddin, M.J.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Prostaglandin G/H synthase 2 | 587 | Mus musculus | Mutation(s): 0 Gene Names: Ptgs2, Cox-2, Cox2, Pghs-b, Tis10 EC: 1.14.99.1 | 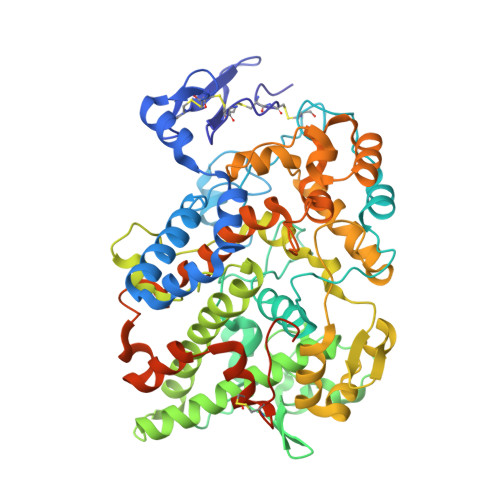 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q05769 | ||||
Glycosylation | |||||
| Glycosylation Sites: 3 | Go to GlyGen: Q05769-1 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| anti-cox-2 alpaca nanobody F9 | 140 | Vicugna pacos | Mutation(s): 0 | 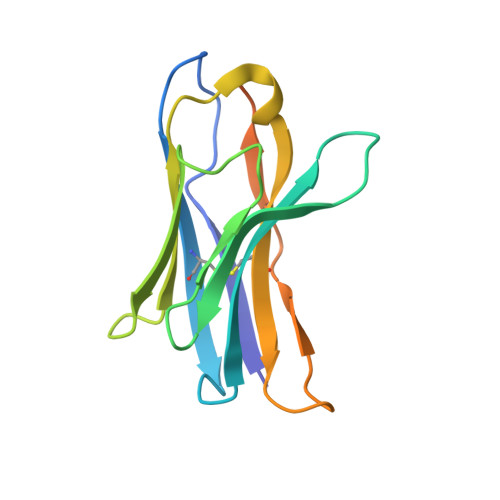 | |
| Ligands 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| NAG Download:Ideal Coordinates CCD File | G [auth A], H [auth A], J [auth B], K [auth B] | 2-acetamido-2-deoxy-beta-D-glucopyranose C8 H15 N O6 OVRNDRQMDRJTHS-FMDGEEDCSA-N |  | ||
| IBP Download:Ideal Coordinates CCD File | I [auth A], L [auth B] | IBUPROFEN C13 H18 O2 HEFNNWSXXWATRW-JTQLQIEISA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 141.526 | α = 90 |
| b = 141.526 | β = 90 |
| c = 90.429 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| Aimless | data scaling |
| PDB_EXTRACT | data extraction |
| XDS | data reduction |
| PHENIX | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| National Institutes of Health/National Institute of General Medical Sciences (NIH/NIGMS) | United States | GM15431 |
| National Institutes of Health/National Institute of General Medical Sciences (NIH/NIGMS) | United States | CA089450 |